Last week, FDA Commissioner Marty Makary resigned from his position amid growing tensions over his priorities, including escalating frustration from pro-life advocates over the slow pace of FDA’s abortion pill review.
Attorney Kyle Diamantas has been appointed as acting FDA commissioner while the administration searches for a permanent replacement.
At first, many pro-life advocates were uncertain about the new acting commissioner.
Reports quickly surfaced that Diamantas previously served as legal counsel for Planned Parenthood.
Simultaneously, pro-life leaders reported receiving requests from Diamantas for a conversation.
March for Life President Jennie Bradley shared on X that Diamantas was on the phone with pro-life leaders within a few hours of being named as the acting commissioner.
Lichter told EWTN News that she shared her concerns about the safety of mifepristone with Diamantas and indicated, based on their conversation, she expected to see “real movement” on the mifepristone study by the FDA.
She told EWTN, “I feel really comfortable that he is bringing in strong pro-life commitments and a commitment to transparency and to moving with all deliberate speed to take a close look at mifepristone and then take decisive action based on what that study shows.”
Live Action President Lila Rose also spoke with Diamantas and posted on X, “He told me he regrets work he performed as an outside attorney for Planned Parenthood…. He shared that he was assigned to the case by his law firm, performed work on it, and ultimately regretted his involvement because of his moral opposition to abortion.”
She also posted, “Diamantas told me that reviewing the abortion pill is a top priority for him and the administration.”
Other pro-life organizations, like Students for Life and Susan B. Anthony Pro-Life America, have been less vocal about who is brought in as the acting FDA commissioner and are more concerned about whether he will act quickly to review mifepristone.
A comprehensive review would include scrutiny of the pill’s safety, emergency complications, adverse effects reporting, telehealth prescriptions and mail-order distribution.
Under the Trump administration, pro-life advocates expected the FDA to conduct a comprehensive review of mifepristone — the first drug in a two-pill regimen that results in a chemical abortion.
Under the Biden administration, safeguards meant to protect women were eliminated, including in-person visits, while mail-order distribution was approved.
In light of a safety study released last May, pro-life advocates have been calling on the FDA to conduct a full and comprehensive review of the drug.
The study, conducted by the Ethics and Public Policy Center, revealed that nearly 11% of women who take the abortion pill experienced serious side effects within 40 days, including sepsis, hemorrhaging, infection and emergency room visits. Those numbers are 22 times higher than the FDA’s current label.
For at least six months, Senator Hawley has been calling on the FDA to stop stonewalling the review of mifepristone and reinstate safety protocols.
But instead of carrying out a comprehensive review of the abortion medication, the FDA approved a generic drug for distribution.
In the last year there has been growing pressure for the FDA to complete the promised safety review.
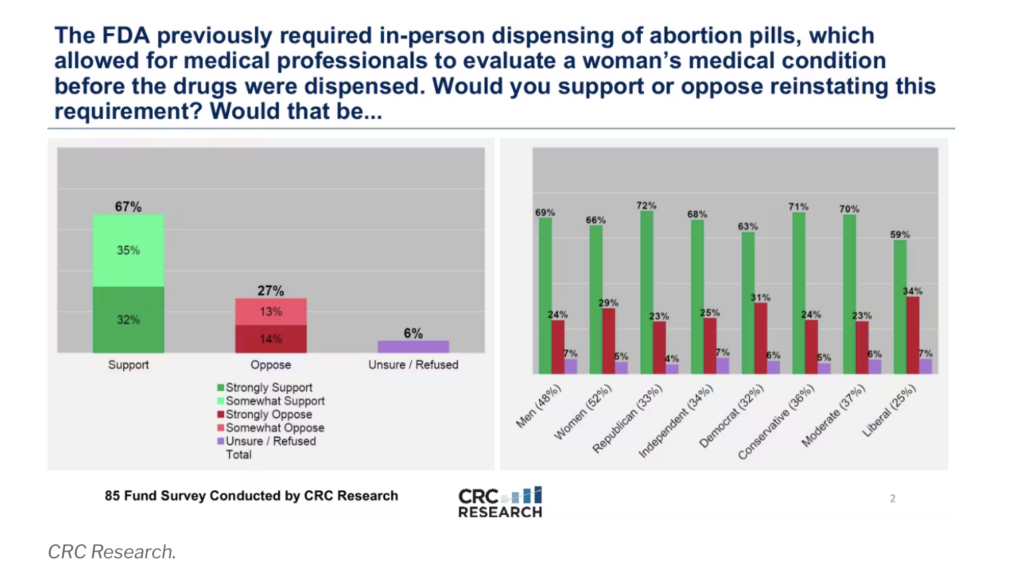
Interestingly, new data shows 70% of Americans, including a majority of each political party, support increased oversight for the distribution and consumption of chemical abortion pills like mifepristone.
Pro-life leaders are calling on Commissioner Diamantas to act now to pull the abortion pill from the market.
The FDA’s review of mifepristone is separate from the legal battle playing out in the federal courts. Judges are still considering questions related to the interstate mailing of abortion pills, while the FDA has the authority to determine drug safety standards.
For now, the pro-life movement seems cautiously optimistic that the FDA transition could create the opportunity for a serious and genuine review of mifepristone.
Some women, after taking the first abortion pill (mifepristone), come to regret their decision. Thankfully, there is a way to reverse the pill’s effects if prompt action is taken.
To learn more about the abortion pill reversal protocol, visit abortionpillreversal.com or call 1-877-558-0333 to be connected with a medical professional who can guide callers through the process of reversing the pill’s effects.
Additionally, if you’re struggling and need a listening ear, Focus on the Family offers a free, one-time counseling consultation with our ministry’s professionally trained counseling staff. To request a counseling consultation, call 1-855-771-HELP (4357) or fill out our Counseling Consultation Request Form.
Related articles and resources:
Pro-Life Groups Raise Alarm Over Delayed FDA Review of Abortion Pill Safety
RFK Jr. Announces ‘Complete Review’ of Abortion Pill After ‘Alarming’ New Study Reveals Dangers
Louisiana, ADF Challenge Biden-Era Abortion-By-Mail Scheme
FDA Approves Generic Abortion Pill Despite Ongoing Safety Review
Become an Option Ultrasound Life Advocate